Essential details
MOQ:1
Product Introduction
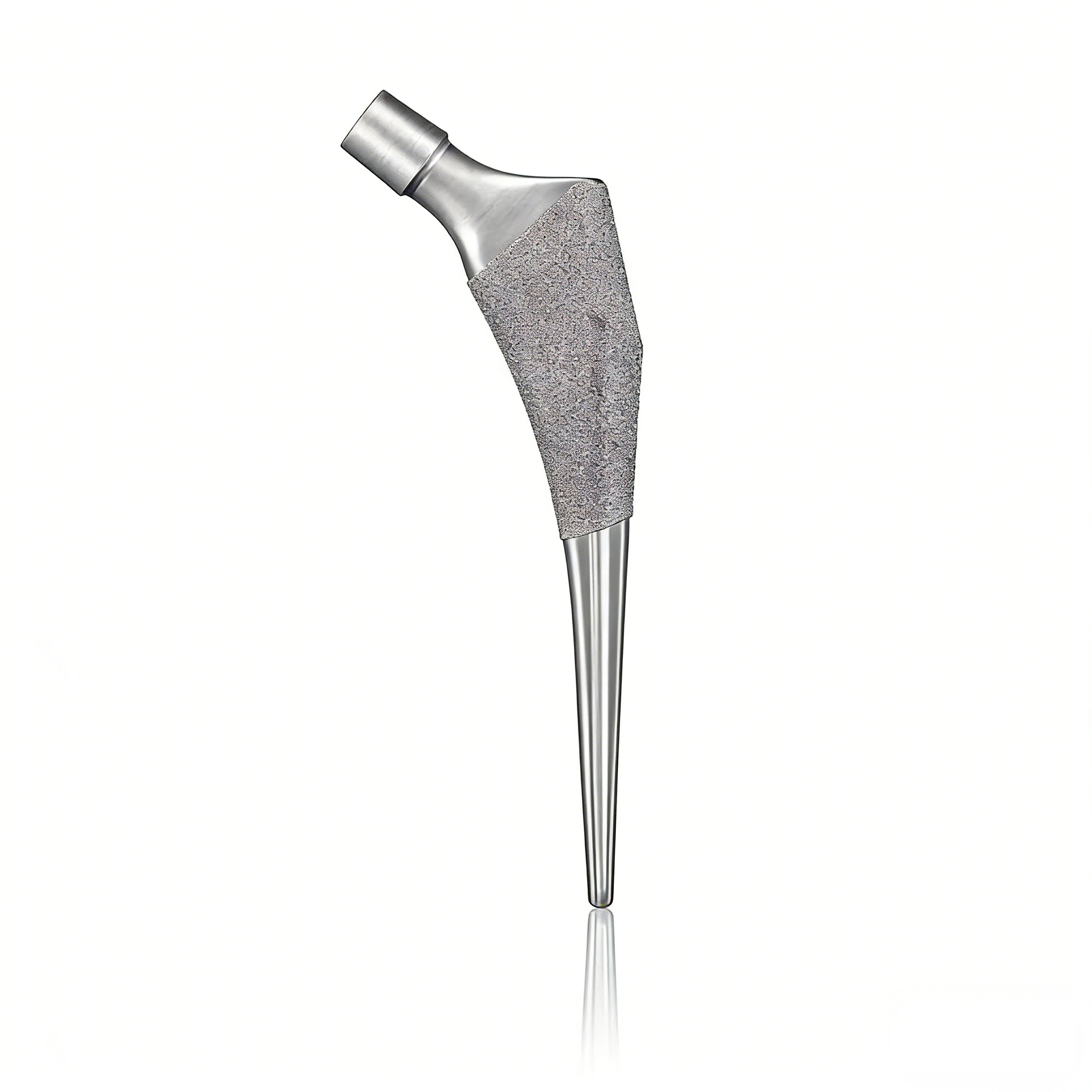
Core FunctionDesigned for total hip arthroplasty, the femoral stem is implanted into the femoral medullary canal to provide long-term stability, transfer biomechanical loads, and achieve reliable osseointegration or cemented fixation.
Material Options
- Ti-6Al-4V / Ti-6Al-4V ELI
- Elastic modulus ~110 GPa, close to natural bone to reduce stress shielding. Ideal for cementless fixation in young, active patients.
- Co-Cr-Mo Alloy (ISO 5832-12)High strength and excellent wear resistance, suitable for cemented stems, revision surgeries and high-load cases.
- 316L Stainless Steel (ISO 5832-1)Cost-effective with stable biocompatibility for economical implant designs.
Optional: 3D-printed porous titanium for enhanced bone ingrowth and primary stability.
Size Specifications
- Length: Extra-short (<90mm), short (90–110mm), standard (110–150mm), long (150–200mm) and extra-long (>200mm) for primary and revision procedures.
- Diameter: 4–20mm with graded sizing to fit different femoral canals.
- Key Geometry: CCD angle 125–135°, offset 38–45mm, tapered/anatomical profiles, with collared or collarless designs for different surgical approaches.
Surface & Coating Technologies
Cementless Stems
- HA Coating: 50–200μm bioactive coating that accelerates bone cell adhesion and osseointegration.
- Porous Coating: 200–500μm for bone ingrowth and strong mechanical interlock.
- Plasma-Sprayed Titanium Coating: Improves surface roughness for stable bone-implant contact.
- Sandblasting: Ra 3–10μm surface finish for mechanical engagement without additional coating.
Cemented Stems
- Polished Surface: Ra <0.2μm to minimize cement abrasion.
- Micro-textured & Phosphatized Finishes: Enhance bonding strength between the stem and bone cement.
Product Classification
- By fixation: Cemented, cementless, and hybrid fixation stems.
- Special types: Revision stems, modular stems, and unicompartmental stems for complex clinical needs.
Key Advantages
- Biomechanically optimized materials reduce stress shielding and ensure long service life.
- Full size range and anatomical designs adapt to diverse patient anatomies.
- Professional surface treatments improve osseointegration and lower loosening risk.
- ISO 13485 certified, strict dimensional control, and fatigue performance meets ASTM F1801.
- Full OEM customization available with low MOQ and efficient lead times.